Autoimmunity, Glucose Precision & The Path to Lifelong Stability
Type 1 diabetes is not caused by sugar (yet not known).
It is not caused by lifestyle (yet not known).
If only we knew the answer to the question “What causes Type 1 diabetes?” Then we might be able to answer, “How can we cure Type 1 diabetes?”
An Autoimmune Condition — Not a Lifestyle Disease

It is not caused by eating sugar.
It is not caused by weight.
It is not caused by poor lifestyle choices.
It is a condition of insulin deficiency — and insulin is essential for life.
Without insulin, survival is measured in days.
With insulin, survival is possible.
With precision, long-term thriving is achievable.
The Historical Turning Point: From Fatal to Manageable
Frederick Banting and his colleagues isolated insulin in the early 1920s — a breakthrough that transformed Type 1 diabetes from a fatal diagnosis into a manageable condition.
Before insulin:
• Children often survived only months
• “Starvation diets” were the only therapy
• Ketoacidosis was inevitable
After insulin:
• Blood glucose could be lowered
• Life expectancy dramatically improved
• Modern diabetes care began
A century later, we now have:
• Continuous glucose monitors (CGMs)
• Rapid-acting and long-acting insulin analogues
• Insulin pumps
• Closed-loop systems
Technology has advanced.
But biology remains the same.
What Actually Happens in Type 1 Diabetes?



-
The immune system becomes misdirected.
-
Immune cells infiltrate the pancreas.
-
Beta cells are gradually destroyed.
-
Insulin production falls to critically low levels.
This destruction may unfold over months or years before diagnosis.
Once approximately 80–100% of beta cells are lost, blood glucose rises sharply — leading to diagnosis, often during diabetic ketoacidosis (DKA).
There is currently no proven prevention.
Genetics play a role, but environmental triggers are believed to initiate the autoimmune cascade.
The Role of Insulin: Why It Matters So Much
Insulin is not just a “sugar hormone.”
It is a master metabolic regulator.
When blood glucose rises after a meal:
-
Beta cells detect the glucose.
-
Insulin is released into the bloodstream.
-
Insulin binds receptors on muscle and fat cells.
-
Glucose transporters (GLUT4) move to the cell surface.
-
Glucose enters the cell for energy or storage.
Without insulin:
• Glucose cannot efficiently enter muscle or fat cells
• Blood sugar rises
• Fat breakdown accelerates
• Ketones accumulate
• DKA risk increases
Insulin is life-sustaining.
Type 1 vs Type 2: The Critical Difference
| Type 1 Diabetes | Type 2 Diabetes |
|---|---|
| Autoimmune | Metabolic |
| Insulin deficient | Insulin resistant |
| Often younger onset | Often later onset |
| Requires insulin immediately | May initially be managed without insulin |
| Usually lean phenotype | Often overweight phenotype |
In Type 1, the body can respond to insulin — it simply cannot produce enough.
In Type 2, the body produces insulin — but tissues resist it.
However, overlap can occur. When insulin resistance develops in someone with Type 1, this is sometimes referred to as “double diabetes.”
Why Is Type 1 So Difficult to Manage?

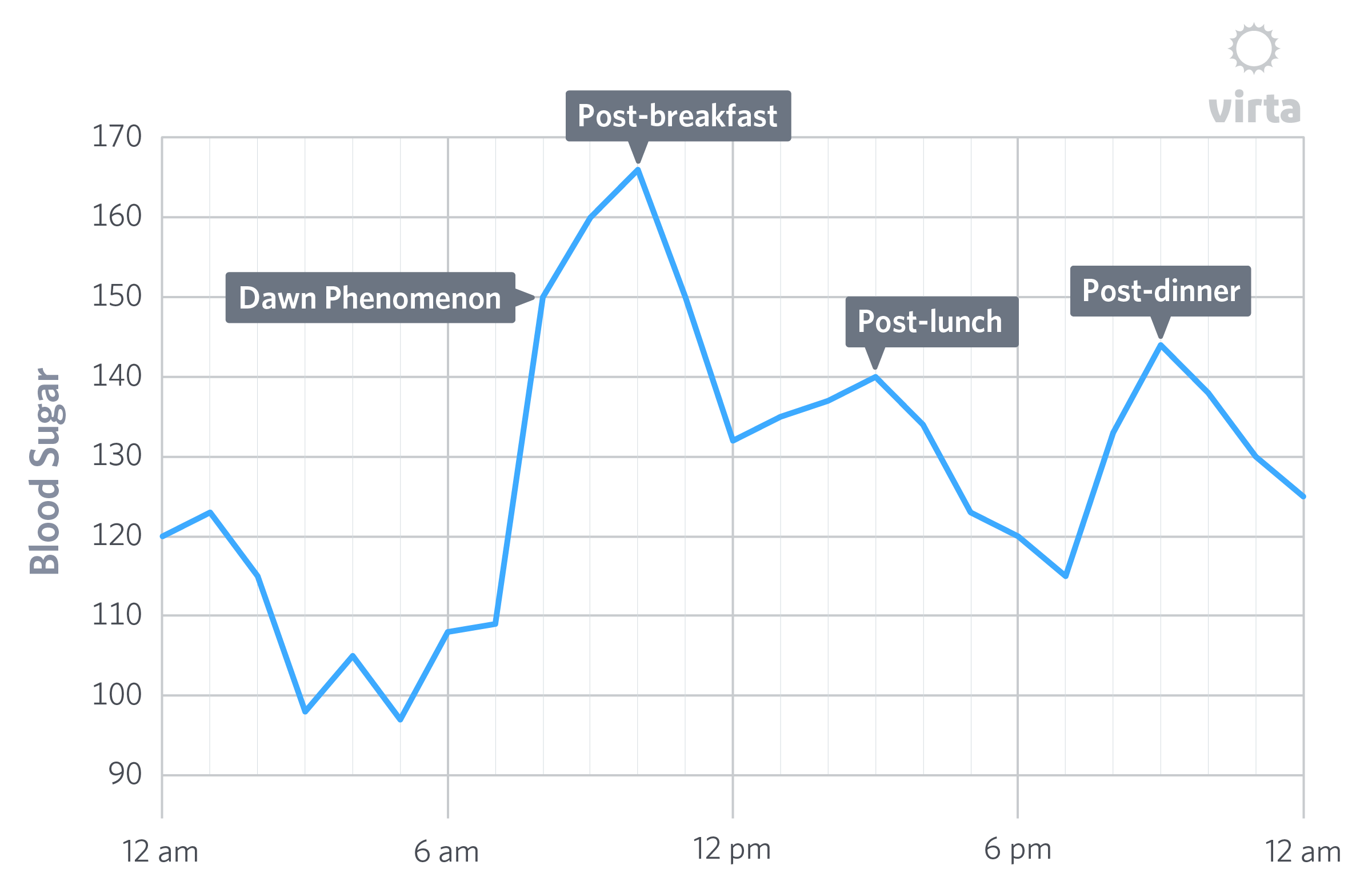

Management is influenced by:
• Sleep quantity and quality
• Growth hormone surges at night
• Dawn phenomenon
• Exercise intensity and timing
• Illness and inflammation
• Stress hormones
• Circadian rhythm
• Meal composition
• Injection site and absorption
• Insulin dosing precision
Even in a fasted state, the liver produces glucose — complicating control further.
The result?
Blood glucose variability — sometimes hourly.
Nutrition: The Largest Modifiable Variable
Food determines how much insulin is required.
Carbohydrates convert to glucose rapidly.
Protein converts more gradually.
Fat has minimal direct glucose impact.
Higher carbohydrate intake → higher insulin doses → greater room for error.
Lower carbohydrate intake → smaller insulin doses → smaller correction margins.
This principle — sometimes called the “law of small numbers” — emphasizes precision and reduced variability.
However, dietary strategy must always be individualized and medically supervised by a low-carb physician, especially in children.
Living With Type 1: Burden and Insight
Type 1 diabetes demands constant decision-making:
• Test
• Calculate
• Dose
• Recheck
• Adjust
For some, this burden is exhausting.
For others — especially researchers and metabolically curious individuals — it offers deep insight into human physiology.
It teaches:
• How sleep alters insulin sensitivity
• How exercise shifts glucose uptake
• How stress hormones affect blood sugar
• How circadian rhythm modifies metabolism
Few conditions provide such real-time metabolic feedback.
The Big Question: Can Someone With Type 1 Live a Normal Life?
With modern care:
Yes — many do.
However, long-term outcomes depend heavily on:
• Glucose stability
• Avoiding chronic hyperglycemia
• Minimizing severe hypoglycemia
• Early complication screening
• Consistent follow-up
Advances in insulin therapy, monitoring, and education have dramatically improved life expectancy compared to the pre-insulin era.
But variability remains a challenge.
Key Takeaways
• Type 1 diabetes is an autoimmune destruction of insulin-producing beta cells.
• Insulin is essential for survival.
• Management requires lifelong insulin replacement.
• Blood glucose variability is influenced by many physiological factors.
• Nutrition plays a major role in insulin demand.
• Modern tools have transformed outcomes — but precision remains critical.
Final Perspective
A century ago, Type 1 diabetes was fatal.
Today, it is manageable — though demanding.
The pancreas may be silent.
But with knowledge, monitoring, and structured care, metabolic stability is possible.
Understanding the biology empowers better decisions.
Education reduces fear.
Precision protects the future.
References
- https://diatribe.org/understanding-diabetes/can-people-type-1-diabetes-also-get-type-2
- https://www.sciencedirect.com/science/article/pii/S016882271630153X
- https://dom-pubs.onlinelibrary.wiley.com/doi/abs/10.1111/dom.13848
- https://www.sciencedirect.com/science/article/abs/pii/S1043276006002633
- https://www.scribd.com/document/920635243/Dr-Bernsteins-Diabetes-Solution
- https://archive.org/details/isbn_9780316182690
- https://www.youtube.com/channel/UCuJ11OJynsvHMsN48LG18Ag
- https://www.youtube.com/watch?v=dPd78PnsQNA
- https://www.youtube.com/watch?v=4X7IwzFPn_Q
- https://www.youtube.com/watch?v=qQc2H26T98E&t=469s
- https://www.youtube.com/watch?v=3xdlzHyysNk&t=7s
- https://www.youtube.com/watch?v=3xdlzHyysNk&list=PLs_TA02I6IvX_FakgvWkfziEciqRSgZnz
- https://www.youtube.com/watch?v=3PZno7Nkuuw
- https://www.youtube.com/watch?v=3xdlzHyysNk&t=7s
- https://www.youtube.com/watch?v=3xdlzHyysNk&list=PLs_TA02I6IvX_FakgvWkfziEciqRSgZnz
- https://www.youtube.com/watch?v=3PZno7Nkuuw
- https://www.youtube.com/watch?v=3xdlzHyysNk&t=7s
- https://www.youtube.com/watch?v=3xdlzHyysNk&list=PLs_TA02I6IvX_FakgvWkfziEciqRSgZnz
- https://www.youtube.com/watch?v=3PZno7Nkuuw
- https://www.youtube.com/watch?v=3xdlzHyysNk&t=7s
- https://www.youtube.com/watch?v=3xdlzHyysNk&list=PLs_TA02I6IvX_FakgvWkfziEciqRSgZnz
- https://www.youtube.com/watch?v=3PZno7Nkuuw
- https://www.youtube.com/watch?v=vyOI9bk3VZc
- https://www.youtube.com/watch?v=um0Ly12Wia8
- https://www.youtube.com/watch?v=zJGAbZIvRh8
- https://www.youtube.com/watch?v=GdmK_SCA2ls
- https://www.youtube.com/watch?v=fA1qh4Sty8g
- https://www.youtube.com/watch?v=zJGAbZIvRh8
- https://www.youtube.com/watch?v=Ho9-oD9KSiw
- https://www.youtube.com/watch?v=3PZno7Nkuuw
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4674464/
- http://diabetes.diabetesjournals.org/content/50/8/1891
- https://www.ncbi.nlm.nih.gov/pubmed/2406181
- http://care.diabetesjournals.org/content/36/12/3860
- https://www.ncbi.nlm.nih.gov/pubmed/10683091
- https://www.ncbi.nlm.nih.gov/pubmed/16036907
- https://www.ncbi.nlm.nih.gov/pubmed/27797909
- https://www.ncbi.nlm.nih.gov/books/NBK278938/table/insulin-pharmacology.factorcomm/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2995531/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3712033/
- https://link.springer.com/chapter/10.1007/978-3-642-84855-1_7
- Abdul-Rasoul, M., Habib, H., & Al-Khouly, M. (2006). ‘The honeymoon phase’ in children with type 1 diabetes mellitus: frequency, duration, and influential factors. Pediatr Diabetes, 7(2), 101-107. doi:10.1111/j.1399-543X.2006.00155.x
- Arora, T., & Taheri, S. (2015). Sleep Optimization and Diabetes Control: A Review of the Literature. Diabetes Ther, 6(4), 425-468. doi:10.1007/s13300-015-0141-z
- Berger, M., Cuppers, H. J., Hegner, H., Jorgens, V., & Berchtold, P. (1982). Absorption kinetics and biologic effects of subcutaneously injected insulin preparations. Diabetes Care, 5(2), 77-91.
- Bluestone, J. A., Herold, K., & Eisenbarth, G. (2010). Genetics, pathogenesis and clinical interventions in type 1 diabetes. Nature, 464(7293), 1293-1300.
- Borghouts, L. B., & Keizer, H. A. (2000). Exercise and insulin sensitivity: a review. Int J Sports Med, 21(1), 1-12. doi:10.1055/s-2000-8847
- Concannon, P., Rich, S. S., & Nepom, G. T. (2009). Genetics of type 1A diabetes. N Engl J Med, 360(16), 1646-1654. doi:10.1056/NEJMra0808284
- The Discovery of Insulin. (2018). Retrieved from http://www.nobelprize.org/educational/medicine/insulin/discovery-insulin.html
- Draper G, D. C., Caughey JL. (1940). The differentiation by constitutional methods between pancreatic diabetes and diabetes of pituitary origin. Trans Am Assoc Phys, 55, 146-153.
- Dublin, L. I. (1951). The Facts of Life from Birth to Death. New York: MacMillan.
- Ferrannini, E., Linde, B., & Faber, O. (1982). Effect of bicycle exercise on insulin absorption and subcutaneous blood flow in the normal subject. Clin Physiol, 2(1), 59-70.
- Filippas-Ntekouan, S., Filippatos, T. D., & Elisaf, M. S. (2018). SGLT2 inhibitors: are they safe? Postgrad Med, 130(1), 72-82. doi:10.1080/00325481.2018.1394152
- Gale, E. A. (2001). The discovery of type 1 diabetes. Diabetes, 50(2), 217-226.
- Gale, E. A. (2009). How to survive diabetes. Diabetologia, 52(4), 559-567. doi:10.1007/s00125-009-1275-1
- Harley, G. (1866). Diabetes, its various forms and different treatments. London: Walton and Mabberley.
- Himsworth, H. P. (1936). Diabetes mellitus: its differentiation into insulin-sensitive and insulin-insensitive types. Lancet, i, 127-130.
- Himsworth, H. P. (1940). Insulin Deficiency and Insulin Inefficiency. Br Med J, 1(4139), 719-722.
- Hinshaw, L., Dalla Man, C., Nandy, D. K., Saad, A., Bharucha, A. E., Levine, J. A., . . . Basu, A. (2013). Diurnal pattern of insulin action in type 1 diabetes: implications for a closed-loop system. Diabetes, 62(7), 2223-2229. doi:10.2337/db12-1759
- Holloszy, J. O. (2005). Exercise-induced increase in muscle insulin sensitivity. J Appl Physiol (1985), 99(1), 338-343. doi:10.1152/japplphysiol.00123.2005
- Jeremy M Berg, J. L. T., Lubert Stryer. (2002). Biochemistry (5th ed.). New York: W H Freeman.
- Johansen, K., Svendsen, P. A., & Lorup, B. (1984). Variations in renal threshold for glucose in Type 1 (insulin-dependent) diabetes mellitus. Diabetologia, 26(3), 180-182.
- Kjobsted, R., Munk-Hansen, N., Birk, J. B., Foretz, M., Viollet, B., Bjornholm, M., . . . Wojtaszewski, J. F. (2017). Enhanced Muscle Insulin Sensitivity After Contraction/Exercise Is Mediated by AMPK. Diabetes, 66(3), 598-612. doi:10.2337/db16-0530
- Lancereaux, E. (1880). Le diabete maigre: ses symptomes, son evolution, son prognostie et son traitement (Vol. 20). Paris.
- Leto, D., & Saltiel, A. R. (2012). Regulation of glucose transport by insulin: traffic control of GLUT4. Nat Rev Mol Cell Biol, 13(6), 383-396. doi:10.1038/nrm3351
- Lister, J., Nash, J., & Ledingham, U. (1951). Constitution and insulin sensitivity in diabetes mellitus. Br Med J, 1(4703), 376-379.
- Maahs, D. M., West, N. A., Lawrence, J. M., & Mayer-Davis, E. J. (2010). Epidemiology of type 1 diabetes. Endocrinol Metab Clin North Am, 39(3), 481-497. doi:10.1016/j.ecl.2010.05.011
- Mazur, A. (2011). Why were “starvation diets” promoted for diabetes in the pre-insulin period? Nutr J, 10, 23. doi:10.1186/1475-2891-10-23
- Permutt, M. A., Wasson, J., & Cox, N. (2005). Genetic epidemiology of diabetes. J Clin Invest, 115(6), 1431-1439. doi:10.1172/JCI24758
- Perriello, G., De Feo, P., Torlone, E., Fanelli, C., Santeusanio, F., Brunetti, P., & Bolli, G. B. (1990). Nocturnal spikes of growth hormone secretion cause the dawn phenomenon in type 1 (insulin-dependent) diabetes mellitus by decreasing hepatic (and extrahepatic) sensitivity to insulin in the absence of insulin waning. Diabetologia, 33(1), 52-59.
- Peter, R., Luzio, S. D., Dunseath, G., Miles, A., Hare, B., Backx, K., . . . Owens, D. R. (2005). Effects of exercise on the absorption of insulin glargine in patients with type 1 diabetes. Diabetes Care, 28(3), 560-565.
- Petersen, K. F., Price, T. B., & Bergeron, R. (2004). Regulation of net hepatic glycogenolysis and gluconeogenesis during exercise: impact of type 1 diabetes. J Clin Endocrinol Metab, 89(9), 4656-4664. doi:10.1210/jc.2004-0408
- Porcellati, F., Lucidi, P., Bolli, G. B., & Fanelli, C. G. (2013). Thirty years of research on the dawn phenomenon: lessons to optimize blood glucose control in diabetes. Diabetes Care, 36(12), 3860-3862. doi:10.2337/dc13-2088
- Redondo, M. J., Fain, P. R., & Eisenbarth, G. S. (2001). Genetics of type 1A diabetes. Recent Prog Horm Res, 56, 69-89.
- Rosenfeld, L. (2002). Insulin: discovery and controversy. Clin Chem, 48(12), 2270-2288.
- Schrezenmeir J., T. F., Tatò S., Laue C., Beyer J. (1993). Differences between Basal and Postprandial Circadian Variation of Insulin Sensitivity in Healthy Subjects and Type 1 Diabetics. Berlin, Heidelberg: Springer.
- Schuit, F. C., Huypens, P., Heimberg, H., & Pipeleers, D. G. (2001). Glucose sensing in pancreatic beta-cells: a model for the study of other glucose-regulated cells in gut, pancreas, and hypothalamus. Diabetes, 50(1), 1-11.
- Takano, A., Haruta, T., Iwata, M., Usui, I., Uno, T., Kawahara, J., . . . Kobayashi, M. (2001). Growth hormone induces cellular insulin resistance by uncoupling phosphatidylinositol 3-kinase and its downstream signals in 3T3-L1 adipocytes. Diabetes, 50(8), 1891-1900.
- Thorens, B., Sarkar, H. K., Kaback, H. R., & Lodish, H. F. (1988). Cloning and functional expression in bacteria of a novel glucose transporter present in liver, intestine, kidney, and beta-pancreatic islet cells. Cell, 55(2), 281-290.
- Recent Post
